Anti-Tau [pS422] (clone 2H9) - 100 µg
Reference: : 4BDX-1501
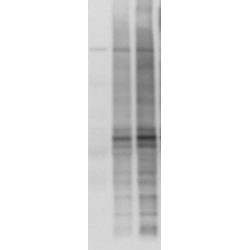
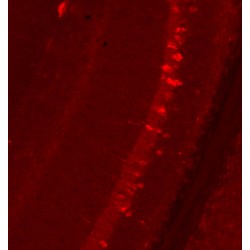
The Anti-Tau antibody pS422 clone 2H9 (4BDX-1501) recognizes the phosphorylated serine 422 of Tau proteins and is suitable for Western Blot, for Immunohistochemistry, Immunofluorescence and Immunotherapy.
This antibody is currently one of the few monoclonal antibodies currently capable of specifically detecting the pathological phospho-epitope of Tau protein in Serine 422.
Data sheet
Applications |
Western Blot , Immunohistochemistry, Immunofluorescence, Immunotherapy |
Reactivity |
Human, Mouse, Rat |
Formulation |
1 mg/mL in PBS |
Conservation |
+4°C / -20°C |
Quantity |
100 µL |
More info
The Anti-Tau antibody clone 2H9 (4BDX-1501) specifically recognises phosphorylated serine 422 of the tau protein (isoform number F; P10636-8: https://www.uniprot.org/uniprot/P10636#P10636-8).
Serine 422 is present on all six isoforms of tau. The 2H9 antibody works by Western blot, Immunohistochemistry (paraffin or frozen sections), Immunofluorescence and even Immunotherapy in mice.
This antibody was generated after immunisation of a mouse with a peptide covering the protein sequence around phospho-serine 422 (Troquier et al., 2012). The antibody is purified from a hybridoma culture supernatant of this mouse. It was selected on its ability to specifically recognise human Tau protein phosphorylated at serine 422 (https://patents.google.com/patent/WO2014096321A1/en).
Phosphorylation of Tau protein at serine 422 is a pathological epitope and thus a marker of neurofibrillary degeneration, one of the major neuropathological features of Alzheimer's disease and tauopathies (Bussière et al., 1999).
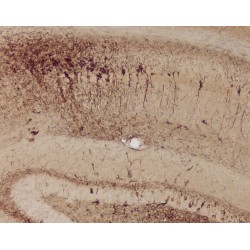
In brain tissue sections, the Anti-Tau pS422 antibody clone 2H9 (4BDX-1501) specifically detects neurons in neurofibrillary degeneration. Thus, in immunohistochemistry, this antibody is an excellent indicator of neurofibrillary tangles in brain tissue sections from Alzheimer's disease patients and in brain sections from mice transgenic for Tau protein.
The Anti-Tau pS422 antibody clone 2H9 (4BDX-1501) also works in immunoblot/western-blot to detect the pS422 epitope in brain homogenates from patients with Tauopathies (Lam et al., 2021), tau experimental models (Sergeant et al., 2019; Tautou et al., 2021).
The pS422 epitope of the Anti-Tau antibody (4BDX-1501) has been targeted for immunotherapy in experimental models of Tauopathies (Troquier et al. 2012; Collin et al. 2014).
The epitope phosphorylated at the serine 422 is found in Alzheimer ‘s diseases (Tolnay et al., 2002) but also in other tauopathies including down syndrome, corticobasal degeneration, progressive supranuclear palsy, pick’s disease and frontotemporal dementia linked to mutation of MAPT gene (Bussière et al., 1999 ; Sergeant et al. 1999 ; Maurage et al., 2004).
The Anti-Tau antibody pS422 clone 2H9 (4BDX-1501) is currently one of the few monoclonal antibodies currently capable of specifically detecting the pathological phospho-epitope of Tau protein in Serine 422.
Recent exemples of use of the Anti-Tau antibody pS422 clone 2H9 (4BDX-1501):
- Lam S, Petit F, Hérard AS, Boluda S, Eddarkaoui S, Guillermier M, The Brainbank Neuro-CEB Neuropathology Network, Buée L, Duyckaerts C, Haïk S, Picq JL, Dhenain M (2021) Amyloid and tau pathologies, cognitive impairments and cerebral atrophy after Alzheimer brain extracts inoculation in a primate. Acta Neuropathol Commun. in press
- Tautou M, Eddarkaoui S, Descamps F, Larchanché P-E, El Bakali J, Goveas LM, Dumoulin M, Lamarre C, Blum D, Buée L, Melnyk P and Sergeant N (2021) A ß-Secretase Modulator Decreases Tau Pathology and Preserves Short-Term Memory ina Mouse Model of Neurofibrillary Degeneration. Front. Pharmacol. Vol 12:679335. doi: 10.3389/fphar.2021.679335
- Lam S, Boluda S, Hérard AS, Petit F, Eddarkaoui S,Cambon K, The Brainbank Neuro-CEB Neuropathology Network, Picq JL, Buee L, Duyckaerts C, Haïk S and Dhenain M (2021) Alzheimer’s brain inoculation in Aß-plaque bearing mice: synaptic loss is linked to tau seeding and low microglial activity. BioRxiv preprint doi: https://doi.org/10.1101/2021.04.06.438654;
- Sergeant N, Vingtdeux V, Eddarkaoui S, Gay M, Evrard C, Le Fur N, Laurent C, Caillierez R, Obriot H, Larchanché PE, Farce A, Coevoet M, Carato P, Kouach M, Descat A, Dallemagne P, Buée-Scherrer V, Blum D, Hamdane M, Buée L, Melnyk P (2019). New piperazine multi-effect drugs prevent neurofibrillary degeneration and amyloid deposition, and preserve memory in animal models of Alzheimer's disease. Neurobiol Dis. Vol. 129:217-233.
To discover of our range of products, click here
Research Use Only Product (RUO) / Not for Diagnosis or Therapeutic Use.